When it entered service in 1983, it was the pride of the Soviet Navy. The Komsomolets, or K-278, was the only one of its kind, with a double titanium hull that allowed it to dive to greater depths than any other submarine. Its plutonium-fueled nuclear propulsion system made it self-sufficient for years. And along with a dozen conventional torpedoes, it carried two nuclear warheads. On April 7, 1989, while sailing in the Norwegian Sea, a fire broke out in compartment 7. The fire spread to nearby compartments through the ventilation pipes, forcing the submarine to surface, only to sink shortly afterward near Bear Island in Svalbard. Forty-two crew members died, most from the frigid water; only 27 survived. Now, at a depth of 1,667 meters, the Komsomolets poses a threat from the bottom of the sea, according to a new study.
“According to one of the first Russian investigations, the nuclear material in the warheads came into contact with seawater due to the physical damage the torpedoes themselves sustained when the Komsomolets sank,” explains Justin Gwynn, a senior scientist at the Norwegian Authority for Radiological and Nuclear Safety. Shortly after the accident, at the height of the Cold War, the Soviets organized several missions using MIR submersibles to assess the condition of the Komsomolets. The Chernobyl disaster had been fresh in their minds, and they needed to allay fears. “This prompted the Russians to cover the cracks on both sides of the torpedo compartment, plug other openings, fill the void in the compartment, and seal the torpedo tubes.” And they did so with titanium.
Gwynn is the lead author of the report on the penultimate mission to the Komsomolets. After the initial Soviet and Russian missions, the Norwegians took over monitoring the sunken submarine. The scientific journal PNAS published the main findings of that operation. The K-278 remains lying on the seabed, buried three meters deep in the sand. Although the structure is still intact, they observed serious damage to the bow and upper deck, specifically in the torpedo compartment. “We found no trace of weapons-grade plutonium in the seawater and sediment samples collected near the submarine’s hull,” Gwynn emphasized.
But during one of the dives, they observed distortions in the water column above the engine compartment’s ventilation pipe. Analyzing this water, they discovered levels hundreds of thousands of times higher than normal. They measured concentrations of 398 kilobecquerels per cubic meter (kBq/m3) of strontium-90 and 792 kBq/m3 of cesium-137. Both this strontium isotope and the cesium isotope are products of the fission of the plutonium and uranium that fueled the Komsomolets’ nuclear reactor, and the becquerel is the unit that measures radioactive activity. These levels exceeded typical radiation in the Norwegian Sea by 400,000 times and 800,000 times, respectively. And that’s despite things having improved considerably since the ship sank.
Regarding the impact on marine life, “in some of the samples of marine organisms we collected on both sides of the submarine, we observed low concentrations of cesium-137, probably due to continuous emissions, but these levels are not expected to have any impact on the organisms themselves,” says Hilde Elise Heldal, a researcher in the department of pollutants and biological hazards at the Norwegian Institute of Marine Research. In fact, she adds in an email: “As you can see in some of the photographs, the submarine’s hull is covered by a thin layer of various marine organisms.”
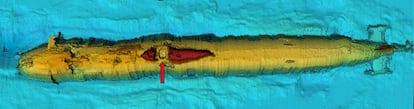
What researchers have also observed is that the reactor fuel is corroding. The uranium or plutonium in nuclear reactors is compressed into cylinders inserted into metal tubes, usually made of zirconium. “If this structure degrades, then the uranium or plutonium is no longer confined,” explains Nuria Casacuberta, a researcher in physical oceanography at the Swiss Federal Institute of Technology in Zurich (ETH Zurich). This scientist uses the presence of radioisotopes in the sea to study ocean dynamics and large ocean currents, so she is very familiar with radioactivity in the sea.
“A small amount of radioactivity still exists in all the oceans, not just on the surface,” Casacuberta points out. And when she says small, she means it: around one becquerel per cubic meter of water on average. “Cesium-137, strontium-90, uranium-235, plutonium-240… these are all elements specific to the nuclear industry; they hardly exist naturally. Any atom we can measure, in almost 99% of cases, is of artificial origin,” explains the researcher, who recently moved to the Institute of Marine Sciences (ICM-CSIC) in Barcelona.
The vast majority of this radiation in the seas comes from nuclear tests conducted in the 1960s and 1970s. Most of these tests were carried out at Bikini Atoll by the United States and in the Arctic archipelago of Novaya Zemlya by the Soviet Union. The second-largest portion of this contamination comes from authorized discharges at the two main nuclear reprocessing plants: La Hague, on the Atlantic coast of France, and Sellafield, on the British coast. A third source is radiation that escaped from the Komsomolets and two other Soviet submarines sunk in the Russian Arctic after reaching the end of their operational life. This calculation does not include the Kursk, another nuclear submarine that sunk in 2000, with the loss of 118 personnel.
It remains to be seen what will happen to the two torpedoes. “They sealed them with titanium plates, and they’ve taken measurements there, and apparently the seal is still working,” Casacuberta notes. “We can’t speculate on whether anything can be recovered. What we can say for sure is that we haven’t observed any indication of weapons-grade plutonium in the marine environment around the submarine,” Gwynn concludes. Even so, they will continue going down to monitor the aging Soviet submarine.
Sign up for our weekly newsletter to get more English-language news coverage from EL PAÍS USA Edition
